
biopharmpt.com
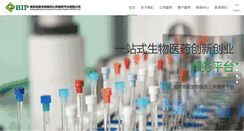
南京高新生物医药公共服务平台有限公司XX,XX化工,产品,电力设备及材料,油田化学添加剂,氟硅新材料,建筑化学材料,特种功能材料,防腐新材料,织物及皮革助剂
http://www.biopharmpt.com/

XX,XX化工,产品,电力设备及材料,油田化学添加剂,氟硅新材料,建筑化学材料,特种功能材料,防腐新材料,织物及皮革助剂
http://www.biopharmpt.com/
TODAY'S RATING
>1,000,000
Date Range
HIGHEST TRAFFIC ON
Wednesday

LOAD TIME
6.7 seconds
16x16
32x32
Nanjing HTZ Biopharmaceutical Public Platform Co.Ltd
zhu yongping
Nanjinggaoxindistri●●●●●●●●●●●●●●●●●●●gyebaitaishengwulou
Na●●ng , Jiangsu, 210061
CN
View this contact
Nanjing HTZ Biopharmaceutical Public Platform Co.Ltd
zhu yongping
Nanjinggaoxindistri●●●●●●●●●●●●●●●●●●●gyebaitaishengwulou
Na●●ng , Jiangsu, 210061
CN
View this contact
Nanjing HTZ Biopharmaceutical Public Platform Co.Ltd
zhu yongping
Nanjinggaoxindistri●●●●●●●●●●●●●●●●●●●gyebaitaishengwulou
Na●●ng , Jiangsu, 210061
CN
View this contact
11
YEARS
0
MONTHS
15
DAYS

SHANGHAI MEICHENG TECHNOLOGY INFORMATION DEVELOPMENT CO., LTD.
WHOIS : whois.cndns.com
REFERRED : http://www.cndns.com
PAGES IN
THIS WEBSITE
0
SSL
EXTERNAL LINKS
1
SITE IP
58.240.74.93
LOAD TIME
6.715 sec
SCORE
6.2
南京高新生物医药公共服务平台有限公司 | biopharmpt.com Reviews
https://biopharmpt.com
XX,XX化工,产品,电力设备及材料,油田化学添加剂,氟硅新材料,建筑化学材料,特种功能材料,防腐新材料,织物及皮革助剂
Biopharmpr.com
The domain biopharmpr.com has expired. If you registered this domain name as a direct customer of Melbourne IT, please click here. To renew your domain name. If you registered this domain name via a reseller of Melbourne IT, please contact the reseller to renew this domain.
Biopharmpro
Good Science is Good Compliance. Audits and Inspection Readiness. Shadle Consulting offers services forBiotechnology and BioPharmaceutical firms :. We offer expertise and guidance in:. Internal and External Audits. Strategic Planning to Build Quality Systems. Experience with Clinical Trials Materials, Commercial Operations, and more.
Biopharmaceutical cGMP Manufacturing Consulting Services
Our firm delivers strategic biopharmaceutical cGMP manufacturing consulting services for biotherapeutics, vaccines and novel drug-delivery products produced with robust manufacturing bioprocesses at production sites worldwide. Biopharmaceutical cGMP Manufacturing Consulting Services. We provide the following consulting services:. Bioprocess Development, Scale-up and Technology Transfer. Due Diligence and Technology Assessment. 5111 Telegraph Avenue, #116. Oakland, CA 94609. NEW EAST COAST OFFICE.
Home « Biopharm Project Solutions Inc.
Process Project Management Compliance. Process and Project Review. Commissioning & Qualification. Manufacturing Process Science and Technology. Project Planning and Management. Media/Buffer Preparation and Use. Cleaning Development and Validation. Malvern, PA, 19355.
Biopharm Quality Assurance Profession
Biopharm Quality Assurance Profession. Thursday, April 07, 2005. The role of Quality Assurance Specialist in the biotechnology industry. Kriger QA Training: Quality Assurance Program. Http:/ www.kriger.com/. This article has been published by the International Biopharmaceutical Association www.ibpassociation.org. The project is sponsored by KRC CRO and training services ( http:/ www.kriger.com/. And ClinQua CRO ( http:/ www.clinqua.com/. Outsource Your Corporate Training. Typical work activities include :.
404 Page Not Found
We cannot locate the page you're looking for. Please check the address and make sure all letters are lowercased with no spaces.
![]() biopharmrecruitingpartners.com
biopharmrecruitingpartners.com
Biopharm Recruiting Partners
Session start() [ function.session-start. Open(/home/content/35/4185135/tmp/sess 1qbl0gtmdc4i833kjhs2fjeog5, O RDWR) failed: No such file or directory (2) in /home/content/35/4185135/html/index.php. Session start() [ function.session-start. Cannot send session cookie - headers already sent by (output started at /home/content/35/4185135/html/index.php:2) in /home/content/35/4185135/html/index.php. Session start() [ function.session-start. Biopharm Recruiting Partners is a Professional Executive Search.
Global Biopharmaceutical Regulations, Inc. – Helping healthcare remain compassionate, cost effective, capable
Your Partner in Evaluating, Mitigating and Reversing Compliance Risk. We help Life Science Industry comply with various regulatory standards. ISO 9001; ISO 13485 and prepare associated scientific documents. Reduces Costs and Avoids Delays Right Before Market Launch with Continuous Preparation for Inspection Readiness. Audit and Gap Analysis. Prevent Delays with Periodic Check of Internal Processes and Supplier Base. Allocate Resources Judiciously with a Strategic Plan for Regulatory Filing. I am personal...
IIS7
Supporting scientists and clinicians, and helping companies sell into life science laboratories and clinics. That assist decision-making in marketing and sales, planning, innovation and customer support. Market information on the use of research and clinical laboratory techniques, and the laboratories where they are being used. Also identifies qualified market prospects to companies supplying into these fields. Circulating Tumour Cells/ Circulating Nucleic Acids. This global market study profiled 313 exp...